The decline in olfaction observed 10–15 years before the first memory lapses is not a coincidence. It is the result of a specific neurobiological mechanism that has been detailed for the first time by researchers from the German Center for Neurodegenerative Diseases and Ludwig Maximilian University of Munich. Their work sheds light on how the brain begins to deteriorate long before the classic symptoms appear.
## Two Key Brain Regions
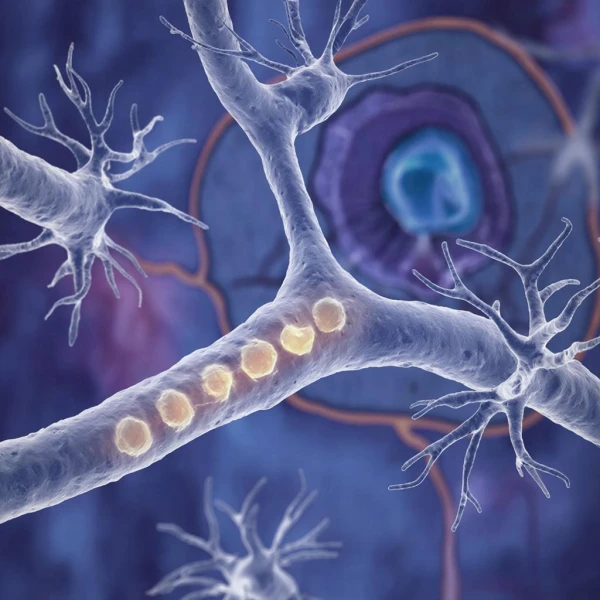
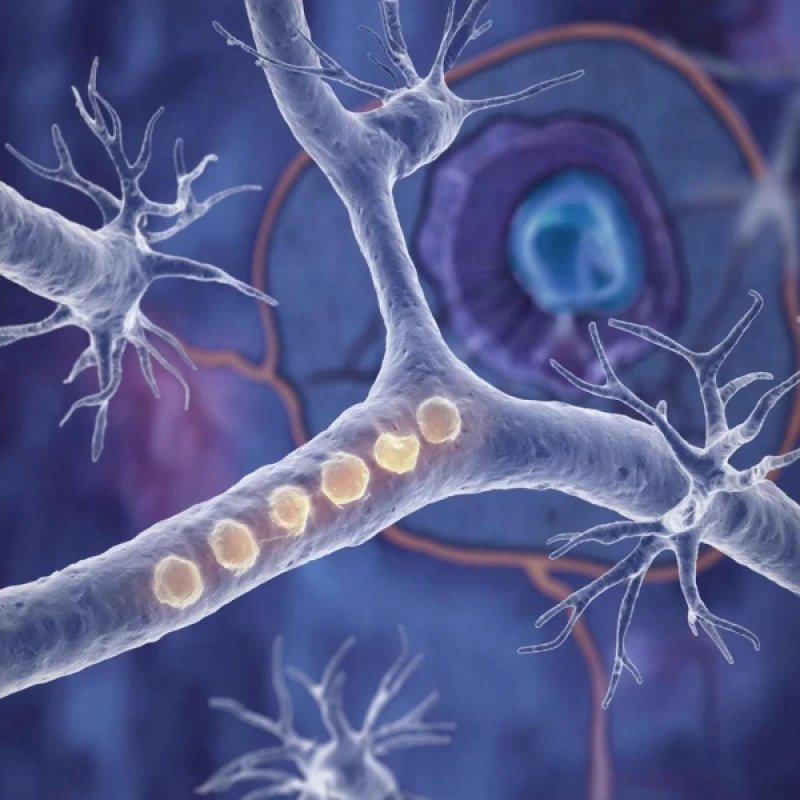
At the core of this mechanism is a complex interaction between two vital structures in the brain: the olfactory bulb and the locus coeruleus. The olfactory bulb, located in the front part of the brain, is responsible for the primary analysis of impulses from the receptors in the nasal cavity.
The locus coeruleus, found in the brainstem, plays a key role in processing sensory signals, including olfactory ones. Its neurons extend long fibers directly to the olfactory bulb. Neurons in the locus coeruleus produce norepinephrine—a neurotransmitter that modulates the excitability of the olfactory bulb neurons and enhances their response to odors. Disruption of this modulation leads to a noticeable decrease in the sensitivity of the entire olfactory system.
## Immune Cells Attack Nerve Fibers
Scientific studies have shown that at the earliest stages of Alzheimer’s disease, specific changes occur in the nerve fibers connecting the locus coeruleus and the olfactory bulb. The molecule phosphatidylserine, normally found inside the neuron membrane, begins to move to its outer side.
The appearance of phosphatidylserine on the outer side of the cell membrane serves as a distress signal that activates the brain’s immune cells. Under normal conditions, this mechanism is necessary for the removal of excess or non-functional neuronal connections, known as synaptic pruning, which is important for brain development and plasticity. However, in Alzheimer’s disease, this process is triggered prematurely and with excessive intensity.
Microglia, recognizing phosphatidylserine and the protein MFG-E8 on the surface of axons, initiate phagocytosis—a process of engulfing and destroying nerve fibers. As a result, norepinephrine projections from the locus coeruleus to the olfactory bulb are gradually destroyed. Experts suggest that the increased activity of damaged neurons during the development of Alzheimer’s disease provokes the movement of phosphatidylserine to the outer side of the membrane.
In other words, neurons begin to generate abnormally frequent electrical impulses, altering the composition of the cell membrane and creating a signal that microglia mistakenly interpret as a command for destruction.
## Evidence from Animals and Humans
This hypothesis is supported by several independent lines of evidence. In experiments on mice with mutations associated with Alzheimer’s disease, researchers observed a loss of 14 percent of norepinephrine fibers in the olfactory bulb by the age of two months. By six months, this figure reached 33 percent.
Alongside the loss of fibers, the animals exhibited a decline in olfaction: they spent 60 percent more time searching for buried food and explored new odors less. Importantly, these changes occurred before the appearance of amyloid plaques—a classic hallmark of Alzheimer’s disease. Genetic suppression of the phagocytic activity of microglia preserved axons and prevented the decline in olfaction, confirming the causal relationship.
Analysis of brain tissue from deceased individuals with early symptoms of Alzheimer’s also revealed degenerative changes in the axons in the area of the locus coeruleus of the olfactory bulb. Positron emission tomography (PET) with the TSPO marker showed increased microglial activity in the olfactory bulb in individuals with the prodromal phase of the disease. These results harmoniously correlate with data obtained from laboratory mouse experiments.
## Clinical Significance and Diagnostic Prospects
Olfactory dysfunction is observed in approximately 85 percent of patients in the early stages of Alzheimer’s disease. Even before the onset of cognitive symptoms, many individuals experience a decline in their ability to distinguish and identify odors, although their sensitivity to them may remain. A large-scale 15-year study involving 1,868 elderly individuals showed that elevated levels of Alzheimer’s disease biomarkers in the blood are consistently associated with a more rapid decline in olfaction.
Participants with the highest concentrations of these markers demonstrated the sharpest decline in olfactory function over the observation period. The practical application of this discovery could be realized through simple and inexpensive olfactory tests. Researchers from Columbia University have shown that a combination of a brief odor identification test with a simple cognitive screening is comparable in accuracy for predicting future declines in cognitive functions and the development of Alzheimer’s disease to costly PET imaging of amyloid deposits.