An international team of researchers has made a significant breakthrough in the production of Doxorubicin, a widely used chemotherapy drug. The study identifies and resolves several molecular “bottlenecks” that have limited the natural production of the drug for more than five decades. By overcoming these constraints, researchers have opened the door to more efficient and cost-effective manufacturing.
A Vital Drug in Cancer Treatment
Approved for medical use in the 1970s, Doxorubicin remains a cornerstone therapy for multiple cancers. Doctors commonly prescribe it to treat breast cancer, bladder cancer, lymphomas, and other carcinomas. Each year, more than one million patients worldwide receive this treatment.
However, bacteria naturally produce this important drug in very small quantities. As a result, pharmaceutical companies have relied on complex and expensive semi-synthetic processes to manufacture it at scale.
Researchers Identify Key Production Bottlenecks
In the new study, scientists uncovered several independent factors that limit the formation of Doxorubicin. According to Keith Yamada, PhD, a lead scientist involved in the research at University of Turku in Finland, identifying these barriers was crucial.
He explained that by addressing these bottlenecks, the team used rational strain engineering to develop a more efficient method of drug production. This approach could help meet the growing global demand for the chemotherapy agent.
Global Collaboration Drives the Research
As per the press release, the breakthrough emerged from an extensive international collaboration involving six research laboratories. The participating institutions included University of Turku in Finland, three laboratories in the United States, and two research groups in Leiden.
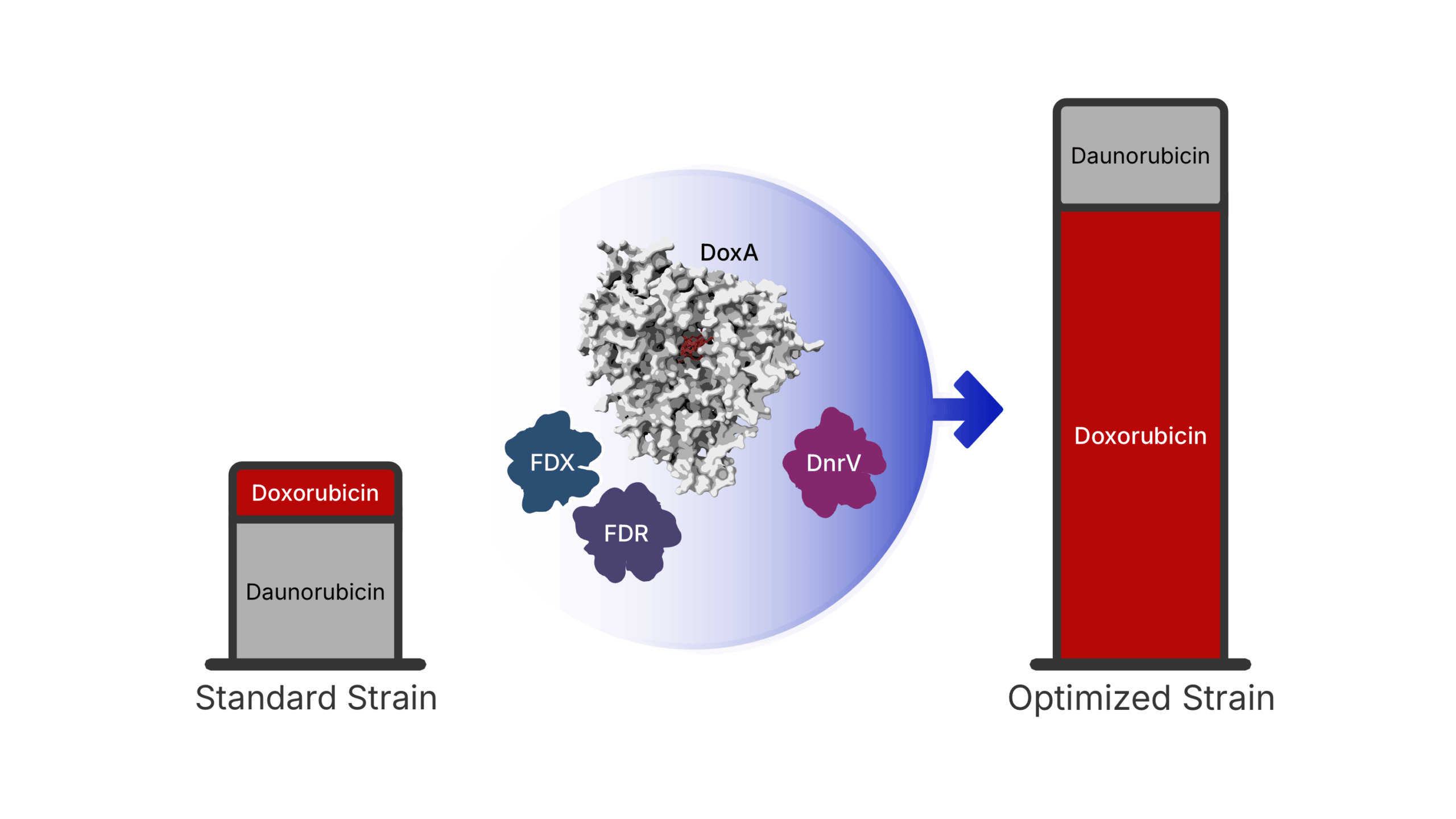
Through this collaborative effort, scientists identified three major constraints that previously prevented high-yield production of the drug.
Discovering the Biological Power Source
First, researchers identified the natural “biological power supply” that drives the drug-producing enzyme. Specifically, they found that redox partner proteins—Fdx4 and FdR3—provide the electron flow required to power the enzymatic reaction responsible for producing Doxorubicin.
By understanding this mechanism, scientists were able to optimize the energy supply that fuels the production process.
Uncovering a Protective Molecular Mechanism
Next, the researchers discovered that a protein known as DnrV acts as a drug-binding “molecular sponge.” This protein binds to Doxorubicin, preventing the compound from interfering with the enzyme that produces it.
Consequently, this protective mechanism ensures that the drug does not shut down its own production pathway inside the bacterial system.
Visualizing the Enzyme Structure
Finally, the team used X-ray crystallography to visualize the enzyme responsible for producing Doxorubicin for the first time. The structural analysis revealed that the drug molecule sits in an unfavorable position within the enzyme, which explains the slow reaction rate observed in natural systems.
This discovery provided critical insight into how the production process could be improved.
Engineering a High-Yield Bacterial Strain
After combining these discoveries, the scientists engineered a new bacterial strain capable of producing 180 percent more doxorubicin than current industrial standards. This improvement represents a major step toward more efficient and sustainable drug manufacturing.
Toward Sustainable Production of Life-Saving Medicines
To translate these findings into practical applications, researchers established the spin-out company Meta-Cells Oy at the University of Turku. The company aims to commercialize these advanced technologies and develop sustainable biosynthetic production methods for essential antibiotics and anti-cancer drugs.
Ultimately, this shift toward fully biosynthetic manufacturing could create a cleaner and more reliable supply chain for life-saving medicines.
The study detailing these findings was published in the journal Nature Communications, highlighting its significance for future pharmaceutical production.