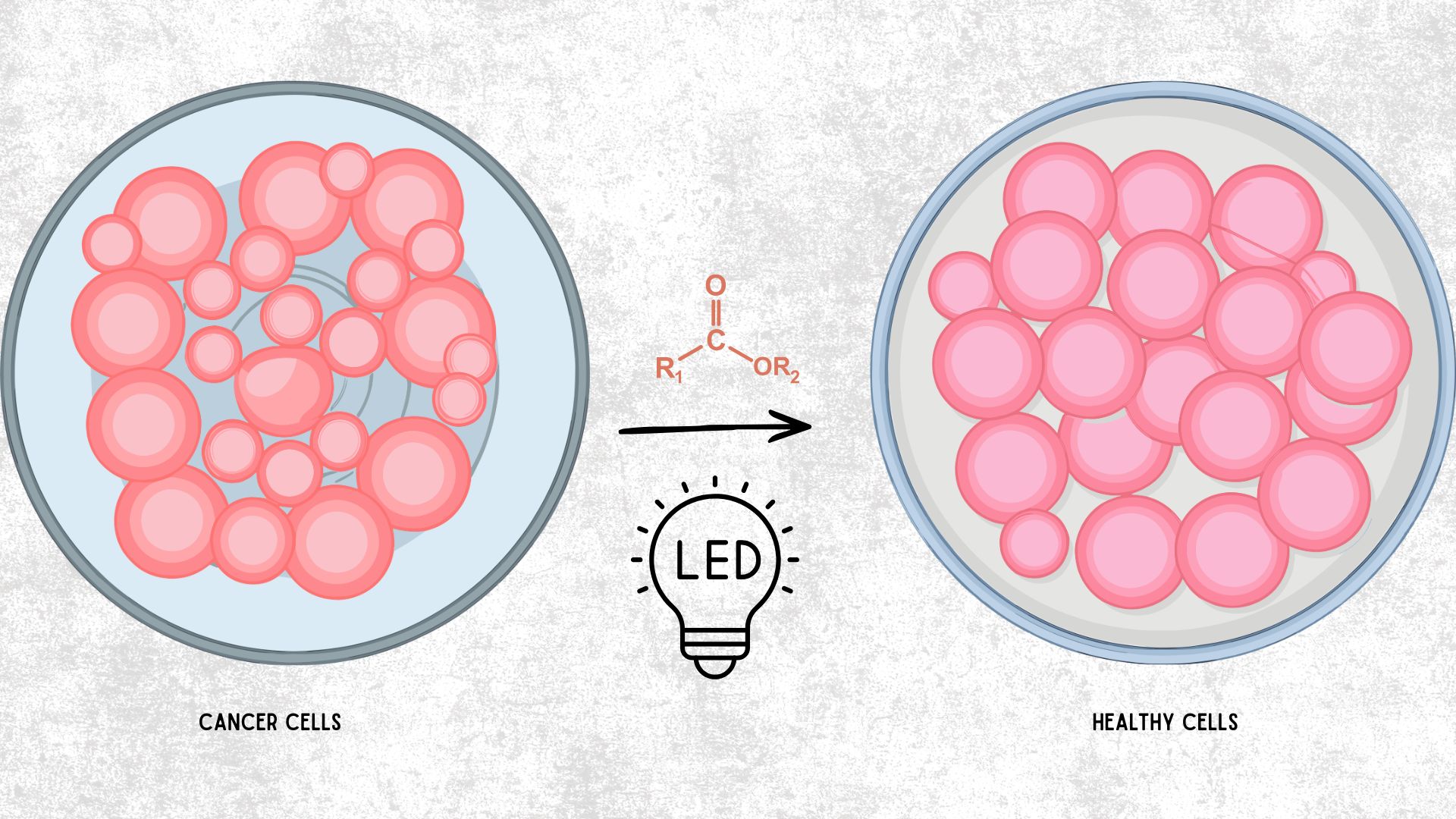
Researchers have synthesized a new family of molecules that halt cancer growth by targeting a specific protein switch. The research focuses on a novel compound known as 14h, which effectively inhibits the Epidermal Growth Factor Receptor (EGFR). This protein often acts as a control lever and triggers cell division in human cancer cell lines, including those for liver, breast, and lung cancers. By blocking this receptor, the new compound forces cancer cells to stop dividing and undergo a programmed self-destruct sequence known as apoptosis.
The research team, including those from the Indian Institute of Science Education and Research (IISER) Pune and the University of Gdansk, Poland, began their search by screening a library of chemical structures called 1,3,4-thiadiazoles. These molecules are considered bio-isosteres, which means they are chemical imposters that can mimic the natural building blocks of life to trick a protein into binding with them. Through an initial screening process, the team identified a molecule that showed promise. They then used computer simulations to model how this molecule fits into the EGFR protein, allowing them to rationally design an even more powerful version.
To synthesise these new compounds, the researchers utilized an environmentally friendly technique called visible-light-driven chemistry. By using blue LED light to trigger the chemical reaction, they were able to efficiently and with high yield build the molecules. This green approach avoids some of the harsh chemicals and expensive metal catalysts often required in traditional drug manufacturing. Once the optimized compound, 14h, was created, it was tested against both wild-type (normal) and mutated versions of the EGFR protein. The results showed that the molecule successfully jammed the protein’s machinery, particularly in liver cancer cells. It also showed remarkable selectivity, meaning it was much less toxic to healthy cells compared to the cancer cells it targeted.
The efficiency of 14h lies in its ability to form a complex network of hydrogen bonds within the pocket of the EGFR protein. Using molecular docking and dynamics simulations, the researchers observed that 14h acts like a precision-engineered key. It fits into the protein’s active site and forms strong attachments to specific amino acids, effectively locking the receptor in an off position. This prevents a process called phosphorylation, which is the signal the cell needs to start growing uncontrollably. Without this signal, the cancer cell’s internal machinery senses something is wrong and triggers its own death.
The study explores a new chemical space with the thiadiazole-based inhibitors, which are structurally different from current FDA-approved drugs like Erlotinib. They offer a potential alternative for patients whose cancers have become resistant to traditional treatments. However, while 14h is highly effective, it is currently less potent than some leading clinical drugs. Additionally, computer models suggest the compound might interact with certain liver enzymes, which could potentially lead to drug-drug interactions if not refined further.
Despite these hurdles, the research provides a vital blueprint for developing novel drugs and treatments. By combining computer modelling with sustainable chemistry, the team has opened a new door for targeted cancer therapy. This work could improve cancer treatments that are not only more effective against resistant strains but also more selective. It could also provide a way to reduce the harsh side effects often associated with chemotherapy and improve the quality of life for patients.