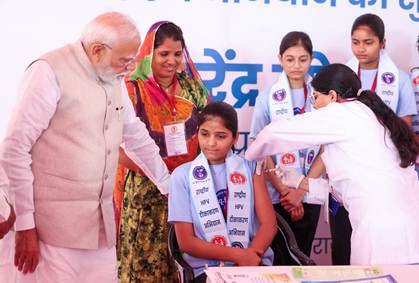
Hon’ble Prime Minister Narendra Modi today launched the nationwide Human Papillomavirus (HPV) Vaccination Campaign for 14-year-old girls from Ajmer, Rajasthan. With this landmark initiative, the Government of India has taken a decisive step toward preventing cervical cancer and strengthening women’s health across the country.
Union Leadership Joins National Launch
Union Health Minister Jagat Prakash Nadda joined the event virtually from Kartavya Bhawan. Smt. Anupriya Patel, Union MoS for Health and Family Welfare, and Dr V K Paul, Member (Health), NITI Aayog, were also present along with senior officials from the Union Health Ministry.
At the same time, leading medical institutions, including AIIMS New Delhi, Safdarjung Hospital, RML Hospital, and Lady Hardinge Medical College, participated in the programme, reflecting nationwide institutional support.
Empowering ‘Nari Shakti’ Through Preventive Healthcare
Addressing the gathering, the Prime Minister described the HPV vaccination campaign as a pivotal initiative to empower India’s ‘Nari Shakti’. He emphasized that when a mother remains healthy, the entire family gains strength and stability. Therefore, the government continues to prioritise women’s health through mission-driven policies.
He cited transformative initiatives such as large-scale sanitation drives, provision of affordable sanitary pads, and the Pradhan Mantri Ujjwala Yojana, which provides clean cooking fuel to millions of households. Additionally, under the safe motherhood scheme, the government transfers ₹5,000 directly into the bank accounts of expectant mothers to support nutritional needs during pregnancy. According to him, these measures reflect a shift toward dignity, care, and sensitivity in women-centric governance.
Cervical Cancer: A Major Public Health Concern
Cervical cancer remains the second most common cancer among women in India. According to the World Health Organization GLOBOCAN 2022 report, India reports over 1.2 lakh new cases and nearly 80,000 deaths annually. Persistent infection with high-risk HPV types, particularly 16 and 18, causes the majority of cervical cancer cases.
Therefore, the nationwide campaign aims to vaccinate girls before potential exposure to HPV infection. The vaccine is safe, effective, and offers long-lasting protection against high-risk HPV strains.
Global Validation and Scientific Backing
Global and national expert bodies, including the World Health Organization and India’s National Technical Advisory Group on Immunization (NTAGI), have recommended HPV vaccination.
Globally, 160 out of 194 countries have introduced the HPV vaccine under their National Immunization Programmes. Notably, 90 countries have adopted a single-dose schedule. India aligns with this global scientific consensus by introducing a single-dose regimen using Gardasil-4 (covering HPV types 6, 11, 16, and 18).
Studies show that HPV vaccines are 93–100% effective in preventing cervical cancer caused by vaccine-covered HPV types. Moreover, high vaccination coverage produces a strong herd effect, thereby indirectly protecting unvaccinated individuals.
Target Population and Coverage
As reported by pib.gov.in, the campaign targets girls aged 14 years (those who have completed 14 but not yet 15 years). According to Registrar General of India (RGI) 2021 estimates, approximately 1.2 crore girls will benefit annually from this initiative.
Operational Framework and Safety Measures
The government has meticulously planned the campaign to ensure safety, logistics efficiency, and effective monitoring.
Key Features Include:
- Campaign Duration: 90 days in campaign mode
- Post-Campaign Availability: Integration into routine immunisation sessions
- Session Sites: Government health facilities such as Ayushman Arogya Mandirs (PHCs), CHCs, SDHs, District Hospitals, and Government Medical Colleges
- Dosage: Single 0.5 ml intramuscular injection in the left upper arm
- Cost: Free and voluntary with mandatory parental/guardian consent
Beneficiaries can pre-register through the U-WIN digital platform or opt for walk-in vaccination at designated government facilities. The system will digitally record parental consent. In areas without internet connectivity, authorities will collect consent in hard copy.
Additionally, the U-WIN platform will manage session planning and reporting, while the e-VIN portal will oversee vaccine stock management and logistics.
Medical Guidelines and Adverse Event Monitoring
The programme strictly follows medical safety protocols. Authorities will defer vaccination in cases of moderate or severe illness, severe allergic reactions to previous vaccines, yeast allergies, or if the girl falls outside the target age group.
For girls who have already received HPV vaccines such as Gardasil, Gardasil-9, Cervarix, or Cervavac, officials will update their vaccination status on U-WIN.
Trained Medical Officers will supervise all vaccination sessions. Furthermore, functional Cold Chain Points (CCPs) and 24×7 government health facilities will remain available to manage any rare Adverse Events Following Immunization (AEFI).
Nationwide Participation and Preventive Healthcare Vision
Following the Prime Minister’s launch in Ajmer, all States and Union Territories organised local rollout events, ensuring simultaneous nationwide implementation.
The nationwide HPV vaccination campaign represents a transformative milestone in India’s preventive healthcare strategy. It reflects evidence-based policymaking, robust preparation, and delivery-focused governance.
The Union Ministry of Health and Family Welfare has urged parents and guardians to ensure that their 14-year-old daughters receive the HPV vaccine during the campaign period. By doing so, families can help secure a healthier, cancer-free future for India’s daughters.