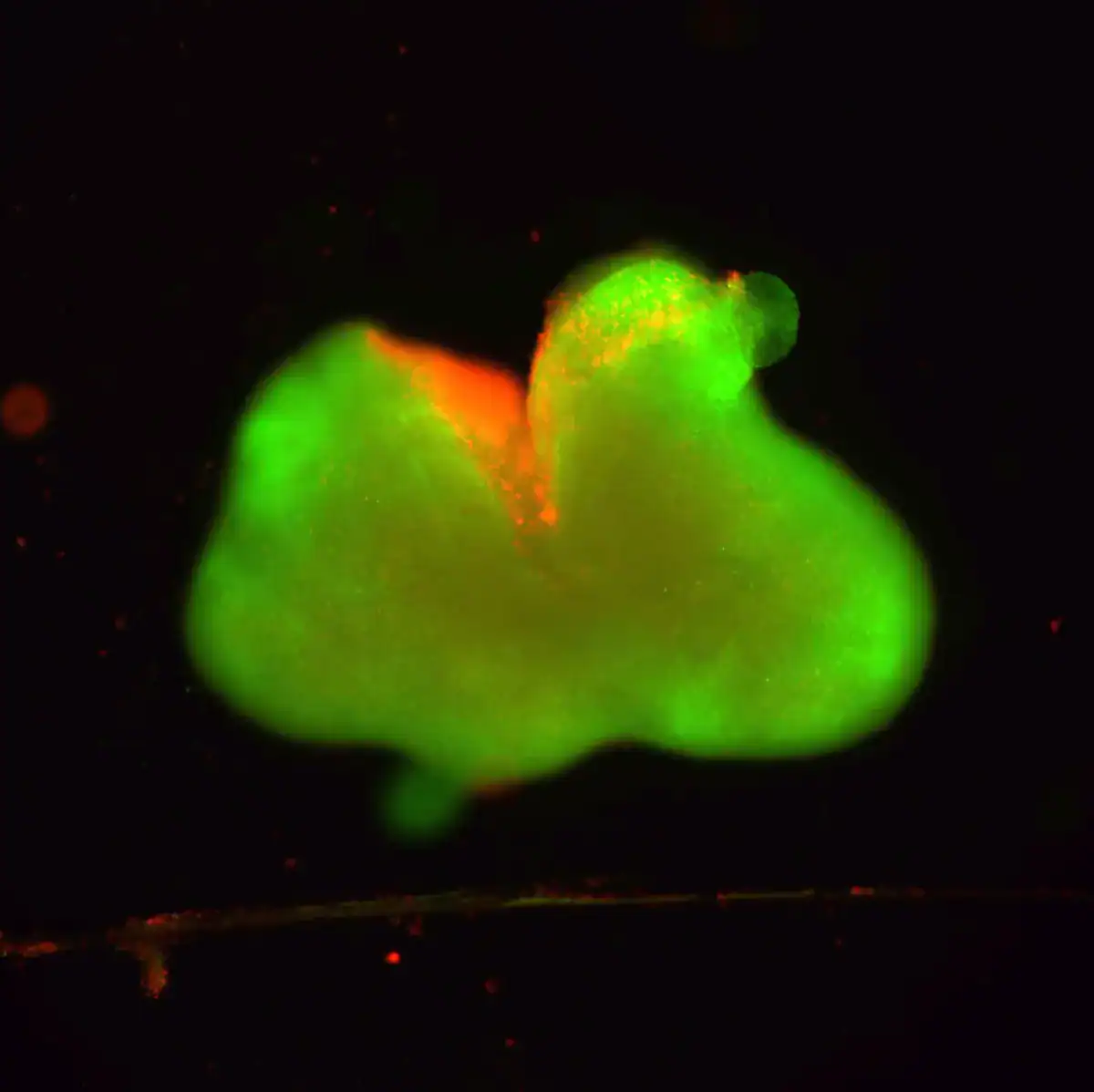
In a petri dish at Northwestern University, researchers grow organoids modeled on the human spinal cord: simplified, lab-made stand-ins built from stem cells. Then they injure them on purpose, either with a scalpel cut or a crushing blow.
The damage sets off a familiar chain reaction. Nerve cells die. Immune signals flare. And eventually a dense barricade of scar-like tissue rises at the wound—one of the main reasons real spinal cord injuries so often lead to lasting paralysis.
Now the team reports that a treatment that once produced dramatic recovery in mice can also push these human organoids toward repair. The approach relies on a gel made from “dancing molecules,” nanofibers designed to move and mingle with cell receptors—an attempt to persuade damaged neurons to extend new connections through hostile terrain.
The new work, published in Nature Biomedical Engineering, offers something new in spinal-cord research: a human model that behaves, in key ways, like an injured spine.
Miniature Injury


Organoids have transformed biomedical research by giving scientists miniature versions of human organs to study in the lab. These tiny structures cannot think, move, or function like full organs, but they can reproduce key cellular behaviors that are otherwise difficult to observe.
Creating a spinal cord organoid is especially challenging. The spinal cord is a complex network of neurons, support cells, and immune cells whose interactions determine whether an injury heals or becomes permanent paralysis.
To build their model, the Northwestern team grew organoids from induced pluripotent stem cells and nurtured them for months until they reached roughly three millimeters in size. By then, the tissue contained neurons, astrocytes, and organized layers resembling those in a developing human spinal cord.
The scientists then produced two common forms of trauma. Some organoids were cut with a scalpel, supposed to simulate penetrating injuries. Others were compressed to simulate the crushing force typical of car crashes or falls.
Both injuries triggered hallmark features of real spinal cord damage: immediate cell death, inflammation, and the formation of glial scars—dense cellular barriers that physically and chemically prevent nerve fibers from regrowing.
The researchers also incorporated microglia, immune cells of the central nervous system, allowing the organoids to generate realistic inflammatory signals. “It means that our organoid has all the chemicals that the resident immune system produces in response to an injury. That makes it a more realistic, accurate model of spinal cord injury,” Samuel Stupp, the study’s senior author, explained in a statement.
Many treatments that succeed in animals fail in humans. A laboratory model built from human cells could help researchers identify promising therapies earlier and discard ineffective ones sooner.
Dancing Molecules


The experimental treatment tested in the organoids is built from peptide molecules that self-assemble into nanofibers, forming a soft scaffold around injured tissue. These molecules carry a biological signal known as IKVAV, derived from proteins that naturally guide nerve growth.
What distinguishes the therapy is motion. The nanofibers are designed so their molecular components shift and rearrange—hence the nickname “dancing molecules.” The movement appears to help the molecules interact more frequently with receptors on nerve cells, stimulating regrowth.
“Given that cells themselves and their receptors are in constant motion, you can imagine that molecules moving more rapidly would encounter these receptors more often,” Stupp said in 2021. “If the molecules are sluggish and not as ‘social,’ they may never come into contact with the cells.”
Earlier animal studies suggested the approach could be powerful. A single injection administered 24 hours after severe injury enabled mice to regain significant walking ability within weeks, according to prior reports.
In the new organoid experiments, the injured organoids treated with the active molecules developed far less scar tissue and showed extensive neurite growth—the thin projections that include axons, which carry signals along the spinal cord.
Even so, major hurdles remain. Real spinal cords contain blood vessels, long-distance neural circuits, and injuries that may be years old rather than freshly inflicted. Treatments that succeed in miniature tissue must still prove safe and effective in the far more complex environment of the human body.
The Northwestern team is already working toward more advanced organoids, including versions that mimic chronic injuries and incorporate additional biological features. Such models could help determine if regeneration is possible and when—and for whom—it might work best.